Many animals exhibit sexual dimorphism









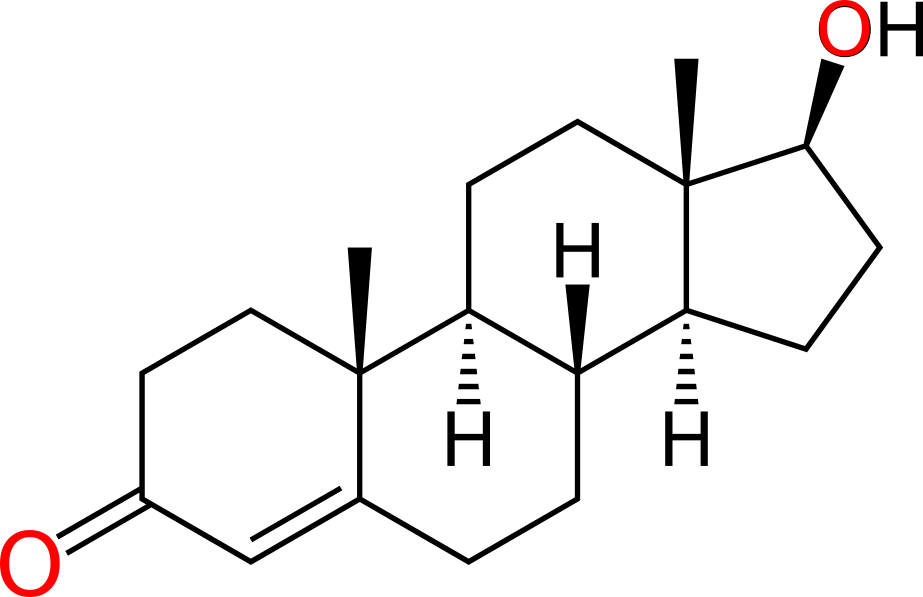
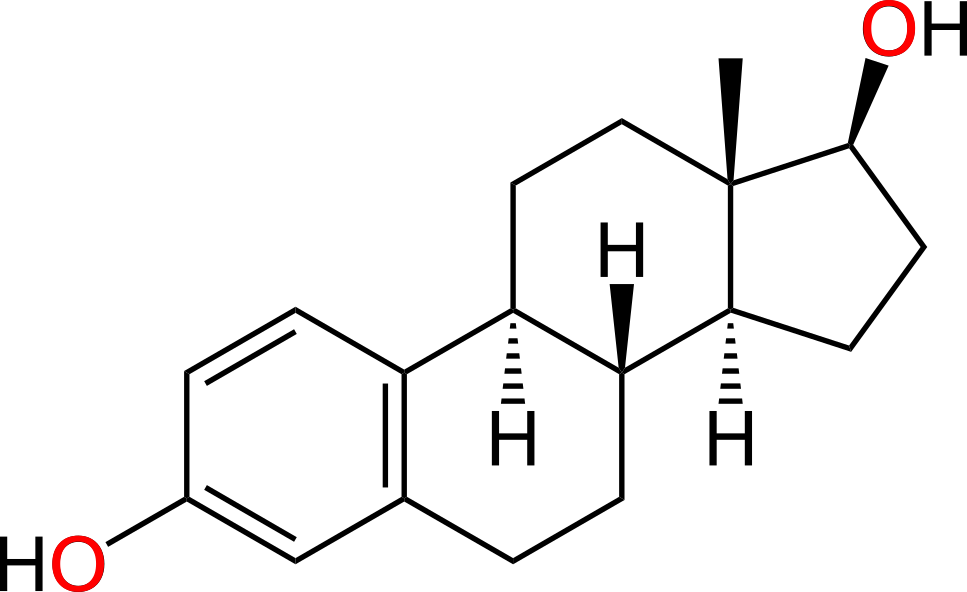

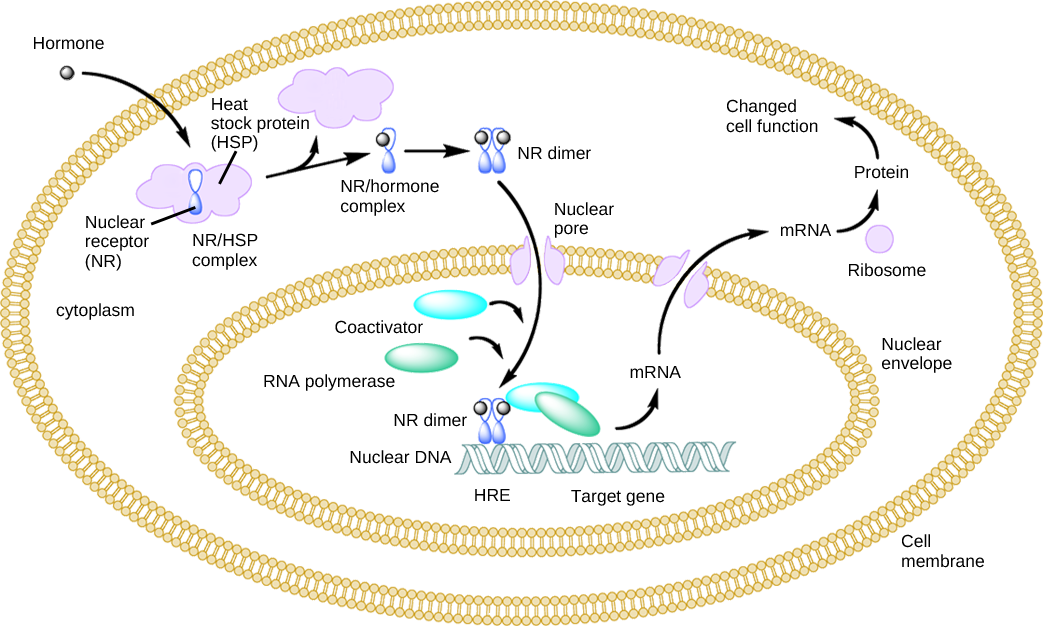
Estrogen and testosterone bind to different receptors, turning on different genes
- Estrogen binds to the estrogen receptor
- Testosterone binds to the androgen receptor
Receptors have almost identical steroid binding pockets
How do almost identical receptors distinguish between almost identical steroids?
Some terms
Affinity: the free energy to bind one steroid.
Specificity: the ability to discriminate between steroids
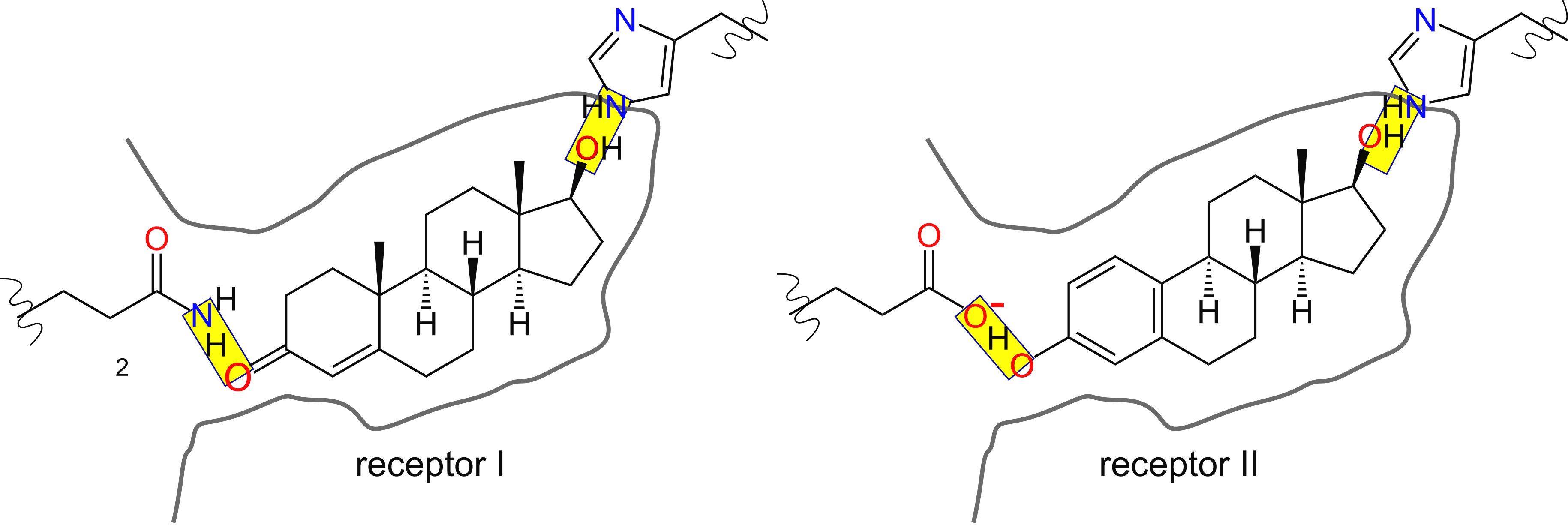
Binding is determined by:
- shape of the pocket
- polar interactions
Biochemists use dissociation constants ($K_{D}$) to measure binding affinity
$ML \rightleftarrows M + L$
$M$ is macromolecule (protein)
$L$ is ligand (small molecule)
We can measure $K_{D}$ by following $\theta$ versus $[L]$
$K_{D} = \frac{[M][L]}{[ML]}$
$[ML] \times K_{D} = [M][L]$
$[ML] = \frac{[M][L]}{K_{D}}$
$\theta = \frac{[ML]}{[M] + [ML]}$
$\theta = \frac{[M][L]/K_{D}}{[M] + [M][L]/K_{D}}$
$\theta = \frac{[L]/K_{D}}{1 + [L]/K_{D}}$
$\theta = \frac{1}{1 + K_{D}/[L]}$
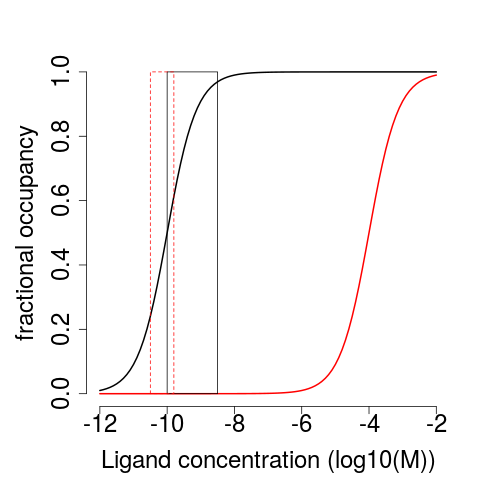
The $K_{D}$ is the concentration of $L$ at which $\theta = 0.5$.
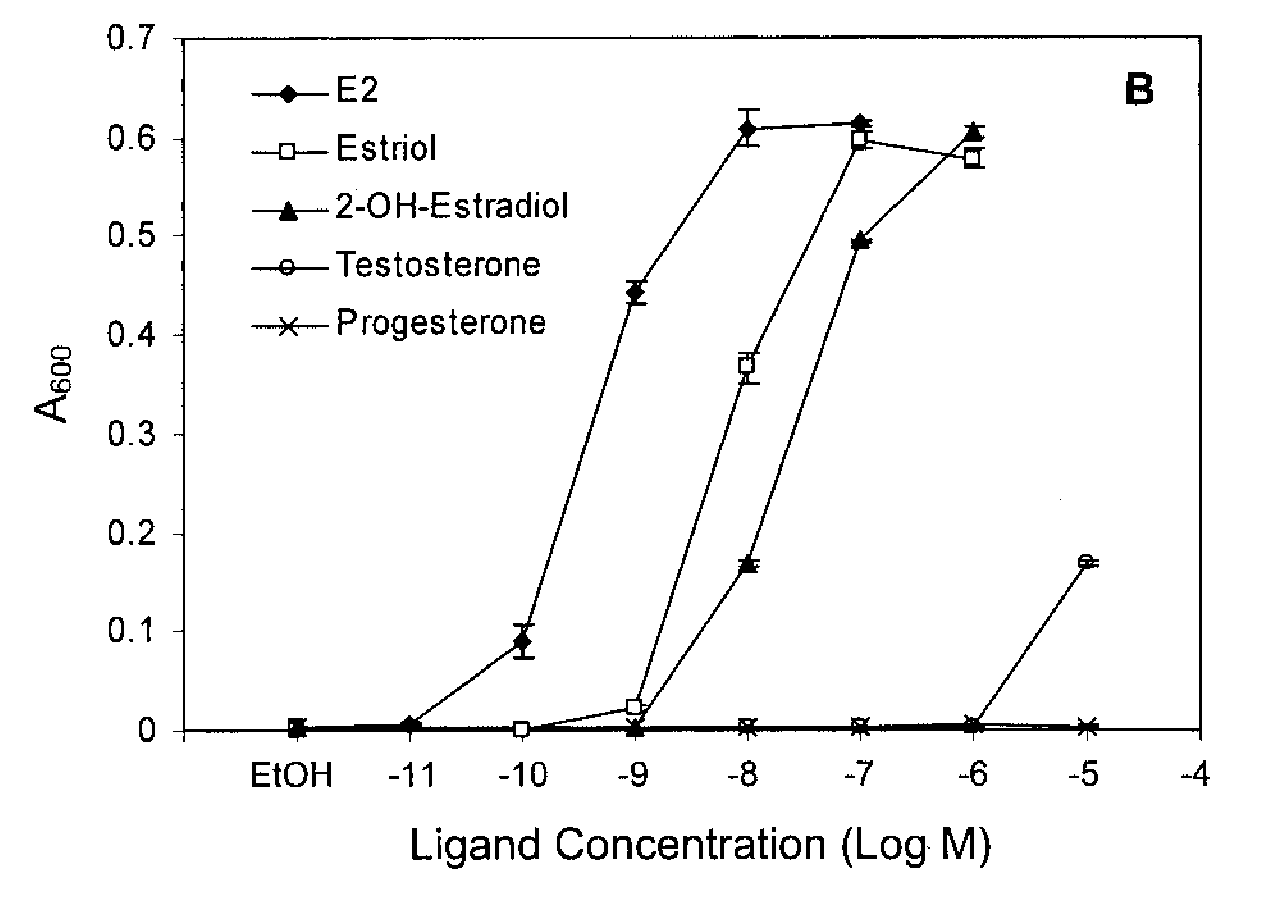 Chen et al. (2004) JBC 279(32):33855-33864
Chen et al. (2004) JBC 279(32):33855-33864
Steroids have only a small range of blood concentrations
ER responds to physiological concentrations of estrogen but not testosterone
Returning to specificity
Is one hydrogen bond, in principle, enough to explain the difference in bindng?
